Introduction
Visual inspection for pharmaceutical manufacturers sits at the center of quality control. Every unit must be examined for defects before reaching patients. Regulatory agencies require manufacturers to demonstrate that defective units are consistently detected and removed, making visual inspection both a patient safety requirement and a fundamental element of GMP compliance.
At the same time, visual inspection is becoming more complex. New container materials, evolving product formulations, and specialized drug formats introduce variability that traditional inspection approaches often struggle to manage consistently. Manufacturers must balance regulatory expectations, production throughput, and inspection accuracy while deciding between manual inspection, semi-automated systems, and fully automated visual inspection technologies.
This report examines how visual inspection systems are used across pharmaceutical manufacturing, the technologies that power them, and hows new AI-based approaches are changing what’s possible.
The Role of Visual Inspection in Pharmaceutical Manufacturing
Product Quality and Patient Safety
The FDA has made its stance clear: visible particulate matter in injectable products can jeopardize patient safety. For pharmaceutical companies, visual inspection requires careful attention because clinical severity depends on the route of administration, the patient population, and the nature of the particulates. The agency expects a complete lifecycle approach to controlling visible particulates, not just end-of-line testing, with preventive controls woven across product development, manufacturing, and ongoing monitoring. A well-run visual inspection program should also reveal upstream issues like equipment wear, environmental contamination, and process drift before they affect entire batches. Vision inspection systems for pharmaceuticals serve as the last line of defense within a broader quality system, and from the FDA’s perspective, they are one piece of a much larger obligation to ensure that only safe products reach patients.
Summary of Regulatory Expectations
The obligation to protect patient safety through product quality is spelled out across multiple layers of regulation, including:
- 21 CFR Part 211
- FDA Guidance on Inspection of Injectable Products for Visible Particulates
- USP 1790
- USP 790
Taken together, these regulations establish a framework that requires 100 percent inspection and AQL testing to continuously verify that the inspection process is effective.
Inspection Points Across Pharmaceutical Production
Pharma visual inspection can occur at three points in the production process: end-of-line, secondary, and upstream. End-of-line inspection is required for all manufacturers. Secondary and upstream inspection may be used depending on the product’s complexity and overall risk profile.

End-of-Line Inspection
End-of-line inspection is the final quality control checkpoint before pharmaceutical products are released for distribution. At this stage, every filled, sealed, and packaged unit is examined to confirm it meets safety and regulatory requirements. Inspection systems look for particulate contamination, verify container closure integrity, and confirm the accuracy of label and packaging information.
Secondary Inspection
Secondary inspection is a follow-up step used to re-evaluate units that were rejected during primary inspection. Typically, the secondary section is performed using manual or semi-automated visual inspection systems, allowing operators to review units previously rejected by automated systems. Confirmed defects are permanently rejected, while units with no identifiable issue may be recovered depending on the company’s quality procedures. This process helps reduce unnecessary product loss and improve manufacturing yield.
Upstream Inspection
In addition to end-of-line and secondary inspection, some manufacturers use upstream inspection to identify problems earlier in the manufacturing process. Detecting defects in near real time allows manufacturers to correct issues quickly and prevent additional defective units from being produced. Upstream inspection is also useful for detecting defects that are difficult to identify later in the manufacturing process, such as container defects and fill-level defects, as well as defects in liquid formulations before lyophilization.
Types of Pharma Visual Inspection Systems
There are three different categories of visual inspection systems available to manufacturers, each offering a different balance of flexibility, throughput, and consistency. The right fit depends on factors such as production volume, product complexity, and a company’s manufacturing lifecycle.
Manual Visual Inspection
Manual visual inspection relies on trained operators who examine pharmaceutical containers by hand under controlled lighting. Inspectors rotate each vial to look for visible particles, cosmetic defects, container damage, and fill level inconsistencies. The strength of this inspection method is in its flexibility. Human inspectors can recognize unusual, never-before-seen defects that traditional automated systems may not be programmed to detect. Manual inspection is best for small batch production or products that are difficult to automate. The major downside of manual inspection is its lack of consistency. Human performance varies from operator to operator and degrades over time from fatigue and attention drift. Throughput is also limited, typically ranging from 5 to 10 units per minute per inspector.

Typical Performance Ranges
- Speed: 5–10 units per minute per inspector
- False reject rate: ~1–3%
- Capital investment: $5K–$25K (inspection booths, lighting, workstations)
- Ongoing investment: Low to moderate
Semi-Automated Visual Inspection
Semi-automated inspection systems combine machine assistance with human judgment. Equipment performs repetitive tasks, such as rotating containers and positioning them under standardized lighting, while an operator evaluates the product and makes the final accept-or-reject decision.
By consistently presenting each product, these inspection machines improve ergonomics and inspection consistency compared to manual methods. Throughput typically increases to 10 to 30 units per minute. However, because human judgment remains part of the process, inspector variability is still a factor. Capital investment generally ranges from $75,000 to $250,000, depending on material-handling requirements, with moderate ongoing labor and maintenance costs.

Typical Performance Ranges
- Speed: 10–30 units per minute
- False reject rate: ~2–5%
- Capital investment: $75K–$250K, depending on system complexity
- Ongoing investment: Low to moderate
Traditional Automated Visual Inspection
Traditional automated visual inspection (AVI) systems use machine vision to detect defects in real time without requiring a human inspector. High-speed AVI is most commonly used for large-volume parenterals packaged in clear vials containing clear liquids. These systems can inspect 200 to 600 or more units per minute, maintain consistent inspection performance across shifts, and generate detailed data that supports quality analysis and traceability.
High-speed AVIs typically cost between $1 million and $4 million and require ongoing maintenance plans. In addition, traditional AVIs require careful algorithm tuning, product-specific validation, and ongoing calibration and technical support. If not properly optimized, these systems can produce high false-reject rates or fail to detect unusual defects that fall outside their trained detection models.

Typical Performance Ranges
- Speed: 200–600+ units per minute
- False reject rate: ~Up to 30% depending on tuning and product type
- Capital investment: $1M–$4M+ for high-speed systems
- Ongoing investment: Moderate to high
Automated Visual Inspection Technologies in Pharma
Automated visual inspection systems are only as effective as the detection technology running inside them. Today, three main approaches power automated visual inspection in pharma, each with a different mechanism for identifying defects.
Rule-based Computer Vision
What is it: Rule-based computer vision systems use predefined rules to analyze measurable image characteristics, such as contrast, edges, size, and brightness. Engineers program specific thresholds, and the system flags products that fall outside those parameters.
Advantages: Results are deterministic and easy to validate in regulated environments. Every decision traces back to a specific rule, which simplifies documentation and regulatory review. These systems also run fast and require minimal computing power, making them well-suited for high-speed inspection lines.
Limitations: The system only detects what it has been explicitly programmed to find. Product variability, lighting shifts, or new container types can require extensive retuning by engineers. This rigidity can lead to a false reject rate exceeding 30% when applied to difficult-to-inspect products with substantial normal variation (lyophilized powders, emulsions, molded glass, etc.).
Supervised Machine Learning
What is it: Supervised machine learning trains models on large labeled datasets of acceptable products and known defect types. The system learns visual patterns from these examples and uses them to classify new inspection images as good or defective.
Advantages: Can detect complex defects that are difficult to define with fixed rules. Because the model learns from labeled examples rather than thresholds, it handles variation in lighting, product orientation, and container appearance better than rule-based approaches. Models can also improve over time through retraining.
Limitations: Models require large volumes of labeled defect images to train effectively (3,000 examples per defect category). In pharmaceutical manufacturing, defects are rare, making it difficult and expensive to build adequate training datasets. New or previously unseen defect types will not be recognized without collecting additional data and retraining the model.
Unsupervised Machine Learning
What is it: Instead of training on defects, unsupervised machine learning builds its model based on an understanding of what a normal product looks like. Once a baseline is established, any unit that falls outside the normal variation within a compliant product is flagged as an anomaly.
Advantages: Unsupervised ML models do not require labeled defect data, which eliminates the biggest bottleneck in deploying AI-based inspection. It can detect previously unseen defect types and subtle quality issues that traditional systems might miss.
Limitations: Since the model does not use labeled defects, it cannot perform defect classification without additional training.
The Gap in Visual Inspection for Difficult-to-Inspect Products
Many pharmaceutical products fall outside the conditions where traditional automated visual inspection (AVI) performs best. Systems designed for clear liquids in transparent containers often struggle with difficult-to-inspect products, where natural variation makes it hard to distinguish between acceptable product characteristics and true defects.
When automation cannot reliably handle these products, manufacturers typically revert to manual visual inspection (MVI) or semi-automated visual inspection (SAVI). While these approaches offer flexibility, they introduce significant limitations. Human inspectors experience fatigue and performance variability, requiring frequent rotation and limiting inspection speeds.
The result is a persistent gap in pharmaceutical visual inspection: products that are too complex for traditional automated systems but produced in volumes too large to be economically handled with manual processes.
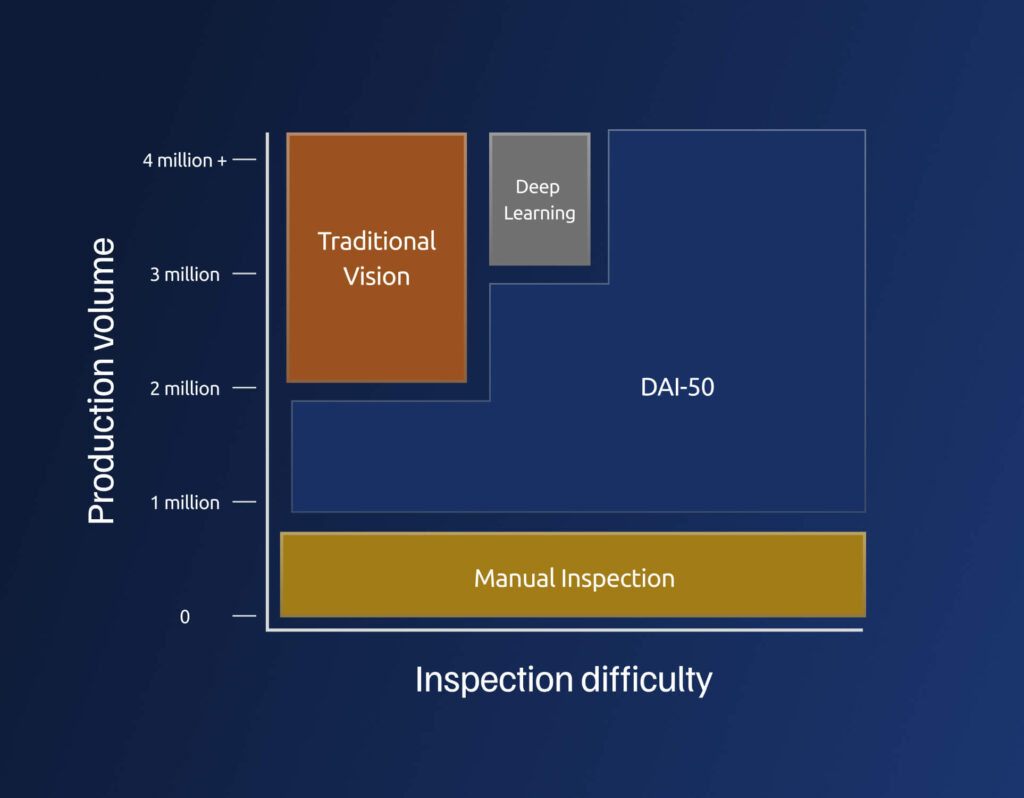
Closing the Gap with the DAI-50
The DAI-50, powered by AVIS, closes this gap by delivering automated visual inspection for vials and syringes with the repeatability of a machine and the flexibility and accuracy of your best human inspector. Using unsupervised machine learning, AVIS models normal product variation directly from 500 pre-inspected compliant units, eliminating the need for labeled defect libraries and enabling detection of both known and previously unseen anomalies. The result is a scalable inspection process that increases throughput, protects yield, and delivers regulatory confidence.















